Our knowledge
Technology
ColiMinder™ measurement approach
Evolving Quality Control through Science
The ColiMinder offers fully automatic and real-time measurements of microbial water quality, enabling a shift from a 2-dimensional process control, based on physical and chemical process feedback, to a 3D control , now involving the microbiological dimension. This works in all water related applications. The incorporation of the microbiological dimension of water quality into process control offers the potential of a drastic increase in efficiency, safety and sustainability of water treatment processes across all industries.
For both economic and security reasons, the possibility of fully automatic process feedback and control for microbial dimension of water quality is a prerequisite for the gradual switch to a circular economy.
Microbiological contamination, most important water quality parameter, still evaluated manually by a 1-3 day lab procedure which is not suitable for automation, online monitoring and process control. ColiMinder technology is based on direct measurement of specific metabolic (enzymatic) activity of target organisms present in the sample.
The technology is extremely robust, directly linked to living organisms and suitable for all applications. Enzymatic activity is scientifically defined exactly
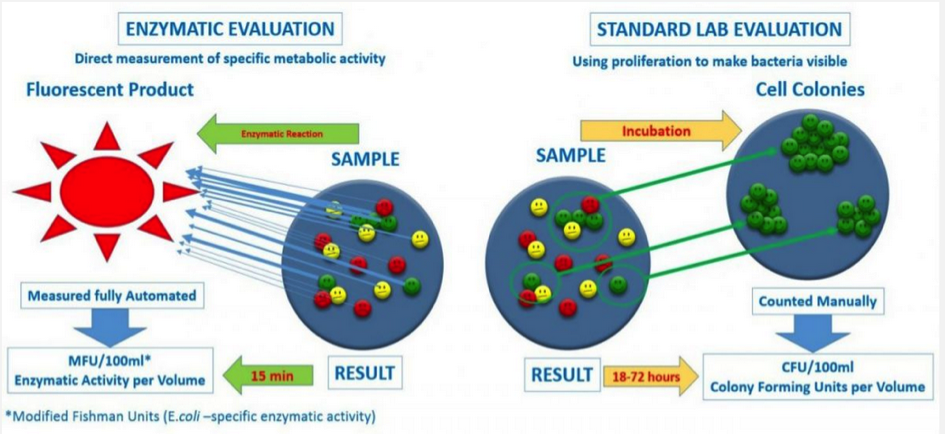
Enzymatic Evaluation
enzymatic approach directly measures the specific enzymatic activity present in the sample. The measured enzymatic activity per sample volume is used as a measure of the contamination.
The enzymatic measurement approach is the only rapid measurement approach that allows
- Technology independent determination if contamination limits
- Calibration of devices independent of their measurement technology
Both conditions are a prerequisite for a future standardization. The enzymatic approach is not limited to water: is it possible to measure other liquids, surface contamination and air/gas.
Standard Lab Evaluation
The classic laboratory testing method uses the proliferation of bacteria to make them visible in order to count them. The number of colonies formed per sample volume is used as a measure of the degree of contamination.
Rapid microbiological measurement approaches
There are two possible basic approaches for rapid measurements of microbiological contamination.
- Examine the individual target organisms
- Measure a signal produced by the total population of target organisms
Traditional Methods
Culture based lab methods are combining both by cultivating individuals to receive a huge number (an amplification of the signal) which can be seen with the naked eye.
Due to the time required for cultivation, culture-based approaches are not suitable for rapid microbiology.
Viability
As per microbiological measurements, it is very important that the technology can distinguish between viable and dead organisms. This is necessary to measure the effects of disinfection or other treatment steps.
In order to measure viability, some time and reagent is required. This can be done by observing a biochemical reaction (using some kind of reagent) which is only occurring in living organisms.
A second theoretical possibility would be the observation of movements of organisms that clearly demonstrate their viability – the disadvantage of this approach is that no movement does not necessarily indicate a dead organism.
Conclusion: Measuring viability requires some time and reagent.
The enzymatic approach evaluates the level of contamination by measuring a signal from all target organisms in the sample volume. This approach requires no sample pre-treatment and less sophisticated technology and is therefore more robust than approaches evaluating individual organisms.
The most basic indication of a viable organism is its metabolism, since it reflects the energy that the cell takes from the environment. This metabolism takes place through enzymes.
There is no more basic measure of viability than metabolic activity.
There are enzymes that are specific to certain organisms or groups of organisms. This offers the possibility to specifically measure the metabolic activity of the respective target organisms.
The metric is the enzymatic activity per volume of a specific enzyme. It reflects the energetic turnover of the target organisms per volume or in other words the concentration of living target organisms, which represents the degree of contamination.
Indicator Bacteria
By comparing different measurement approaches, it is important to understand the principle of indicator bacteria. E. coli, Enterococci and Total Activity (ALP) are used as indicators of fecal and microbiological contamination. The higher the contamination, the higher the risk of pathogens and all kinds of infectious germs that spread through the metabolic pathway. For this reason, water is generally tested for E. coli and others.
The question to be answered by testing for indicator bacteria is:
How high is the level of fecal contamination of a sample? – because this indicates the level of risk
The classical method to evaluate the level of contamination is utilizing the growth of bacteria (E. coli, Enterococci or HPC) as a measurement instrument, and the number of colonies formed, to determine the level of contamination. The ColiMinder® is using the metabolic activity of target organisms “specific enzymatic activity) present in the sample, as a measure for how many living target organisms are present per sample volume, to determine the level of contamination and risk.
Scientific Definition of enzymatic activity
One important fact may be mentioned here: enzymatic activity, for example the E. coli specific ß-Glucuronidase activity, is scientifically exactly defined.
An exact scientific definition of ß-Glucuronidase is given in Modified Fishman Units:
GUS / E.coli specific enzymatic activity
MFU (Modified Fishman Unit): One MFU will liberate 1.0 μg of phenolphthalein from phenolphthalein glucuronide per hour at pH 6.8 at 37°C.
As a direct consequence VWM´s ColiMinder® is calibrated and delivering a result on E. coli contamination in Modified Fishman Units per Volume, representing E. coli -specific enzymatic activity per volume of sample as a direct measure representing the number of living organisms per sample volume. As the measurement parameter of enzymatic activity is scientifically exactly defined it provides the base to adopt respective limits of tolerable contamination expressed in enzymatic activity, for all kinds of applications.
GLU / Enterococcus specific enzymatic activity :
Beta-D-Glucosidase (EC 3.2.1.21, CAS 9001-22-3) is measured in “GU (Glucosidase Units)”. Unit Definition: One GU (Glucosidase Unit) liberate 1.0 µmol of p-nitrophenol (pNP) from p-nitrophenyl-β-D-glucopyranoside (10 mM) per minute in 50 mM sodium maleate buffer, pH 6.5 at 40°C (5).
ALP / total enzymatic activity:
Alkaline Phosphatase (EC 3.1.3.1, CAS 9001-78-9) is measured in “U (Units)”. Unit Definition: One Unit (U) hydrolyzes 1.0 µmol para-nitrophenyl phosphate (pNPP) per minute in glycine buffer at pH 10.4 at 37°C.
GEt in touch
Feel free to contact us with any query – we are delighted to answer all your questions.
